October 20, 2022
CDC Approves the New Novavax COVID-19 Immunization for Adults
July 18, 2022
Today's Facts About COVID-19
April 24, 2021
COVID-19 vaccines are saving lives, reducing hospitalizations and preventing death
April 7, 2021
COVID-19 Vaccine Ingredients
April 1, 2021
COVID Vaccines Work
October 5, 2020
There is no such thing as a mild covid-19 infection
August 14, 2020
COVID-19 Is Not Just a Cold
July 20, 2020
NEW TREATMENT TO PREVENT COVID-19 INFECTION
Our tax dollars are hard at work, developing new treatmentsto keep all of us healthy.
On July 14, 2020, the New England Journal of Medicinepublished successful results using Moderna’s new treatment to prevent severeCOVID-19 infections. https://www.nejm.org/doi/full/10.1056/NEJMoa2022483
Chief investigator Dr. Lisa Jackson at the Kaiser PermanenteWashington Health Research Institute in Seattle, Washington and hercollaborators stated: “The mRNA1273 vaccine induced anti–SARS-CoV-2 immuneresponses in all participants, and no trial-limiting safety concerns wereidentified. These findings support further development of this vaccine.”
Moderna has been working closely with and funded in part byAmerica’s talented infectious disease experts at the National Institute ofAllergy and Infectious Disease (NIAID) to create a safe and effectiveant-COVID-19 treatment, and thus far it appears to be a success. (NIAID:https://www.niaid.nih.gov/ Moderna: https://www.modernatx.com/
Quoting the article: “We conducted a phase 1,dose-escalation, open-label trial including 45 healthy adults, 18 to 55 yearsof age, who received two vaccinations, 28 days apart, with mRNA-1273 in a doseof 25 μg, 100 μg, or 250 μg. There were 15 participants in each dose group.”Fatigue, chills and localized pain were common, as expected.
As seen below, their results indicate two 100 microgram (100ug) injections of messenger RNA (mRNA) that codes for production of the virus’spike protein will be safe and effective in preventing severe Coronavirus-2infections. Additional studies are underway in three thousand volunteers toconfirm these initial findings, the results of which will be available laterthis year.
Help is on the way…

May 19, 2020
New COVID-19 Vaccine
It was widely reported on May 17, 2020, that Moderna corporation’s new mRNA based vaccine induced anti-viral antibodies to the SARS-CoV-2 coronavirus in 8 volunteers, which may lead to the prevention of life-threatening coronavirus-2 infections. https://www.cnn.com/2020/05/18/health/coronavirus-vaccine-moderna-early-results/index.html
Moderna is working with the National Institute of Health (NIH) in the most important vaccine research in the past century. If future studies go well, the company's vaccine could be available to the public as early as January, Dr. Tal Zaks, Moderna's chief medical officer, told CNN. "This is absolutely good news and news that we think many have been waiting for for quite some time," Zaks said. https://www.cnn.com/2020/05/18/health/coronavirus-vaccine-moderna-early-results/index.html
The CoV-2 virus infects human cells in human respiratory and gastrointestinal systems using ACE2 receptors - as depicted below. This new vaccine may be able to protect humans by neutralizing the virus before it infects its victim.
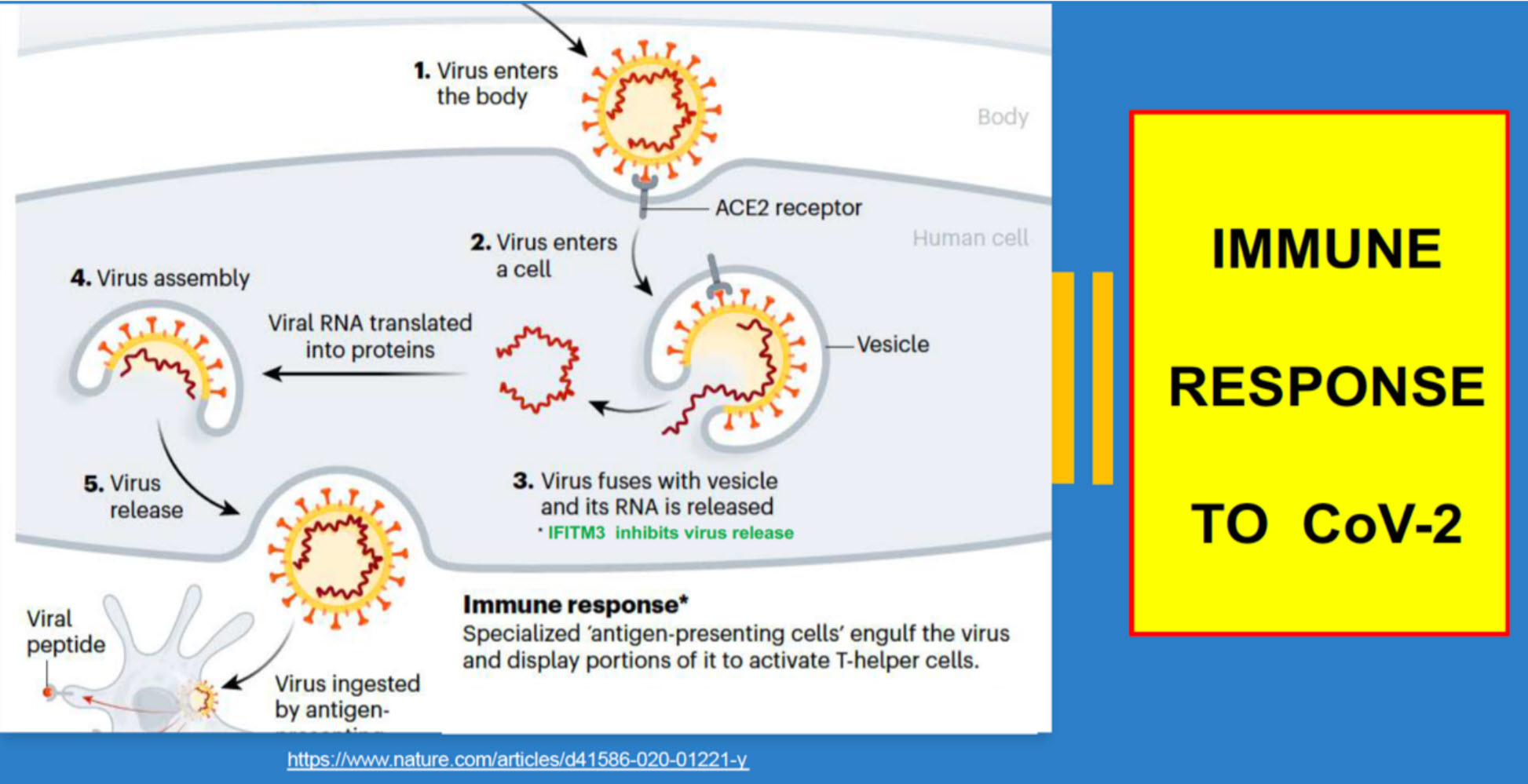
Moderna and the NIH will continue testing this new vaccine in many more volunteers prior to allowing everyone to receive it.So, there is Hope we will overcome the COVID-19 Pandemic by next year.

